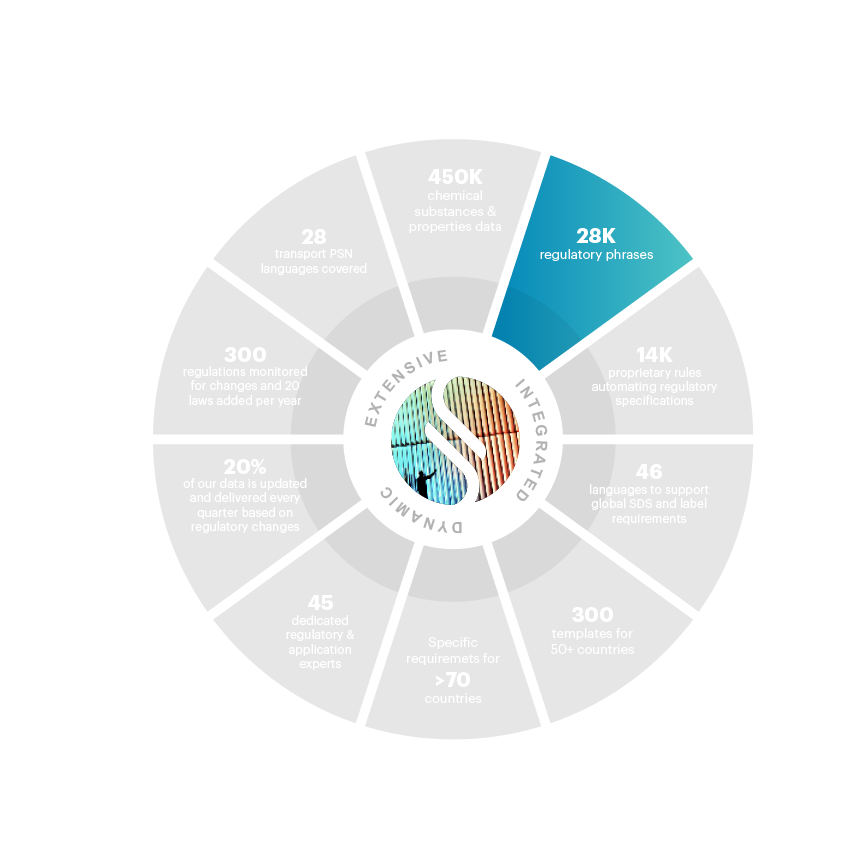
28,000+ phrases for authoring
- Labeling phrases from primary regulatory sources
- Non-regulatory phrases translated and peer reviewed by domain experts
- Exposure Scenarios phrases from ESCOM standard phrase Catalogue
Managed Regulatory Content helps you navigate the complexities of global chemical regulations, so you can comply with confidence.
Request a demoNavigating global chemical regulations demands the right mix of technology and managed regulatory content for compliance. Sphera's authoring solutions offer industry-leading content for confident compliance.
28,000+ phrases for authoring
14,000+ document authoring

46 languages translated

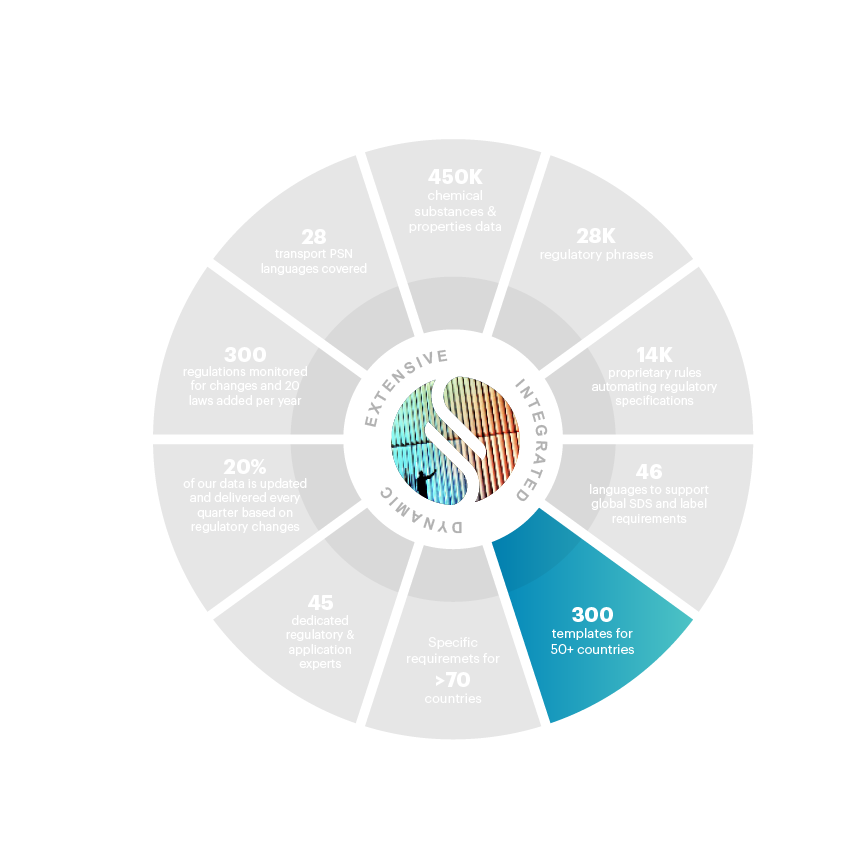
300+ templates provided in the solution
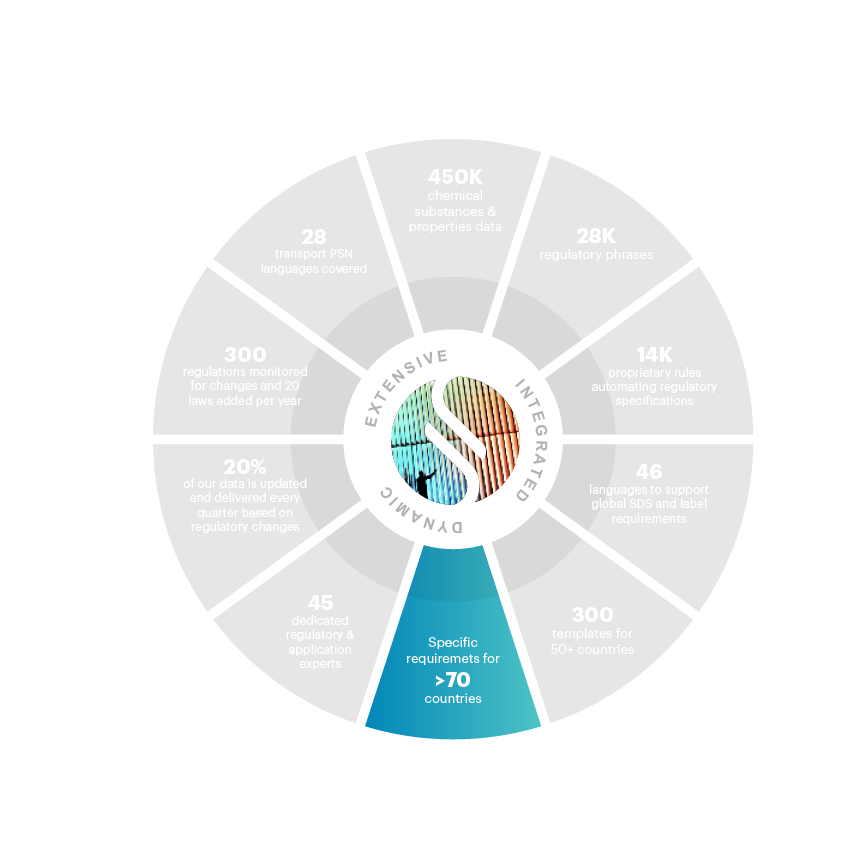
70+ requirements made for countries
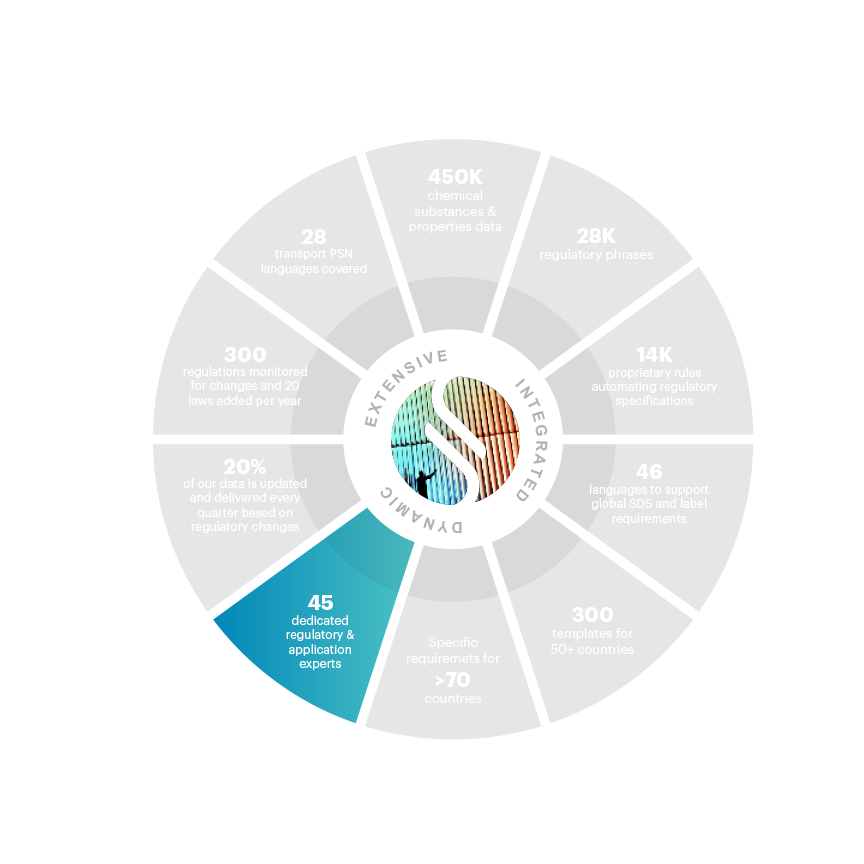
45 full time employees
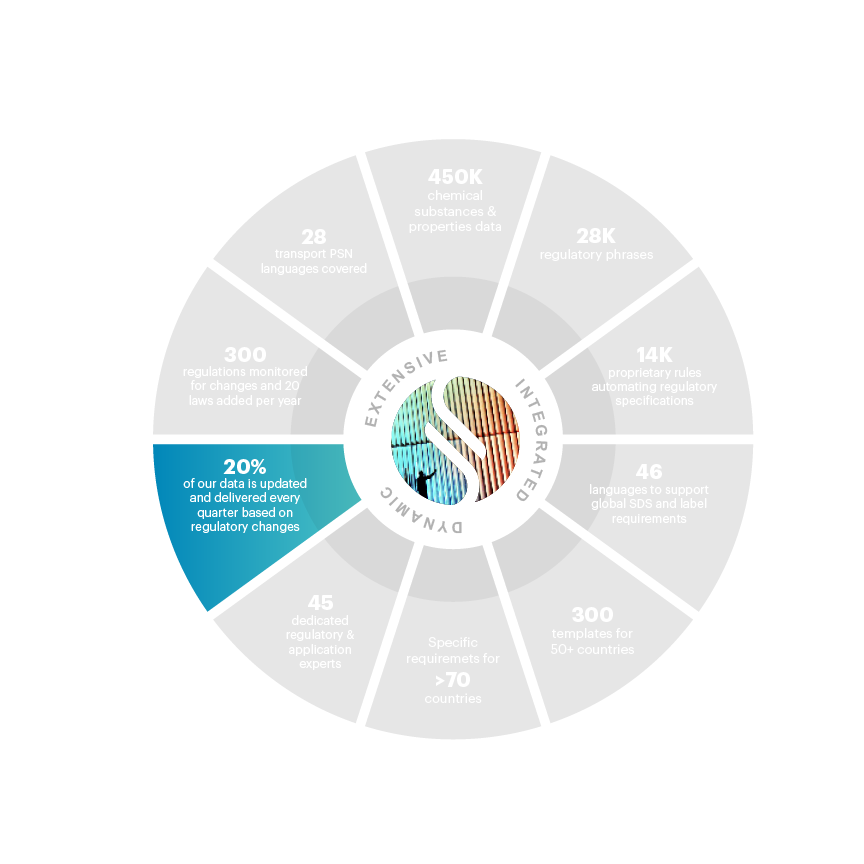
20% updated and delivered quarterly
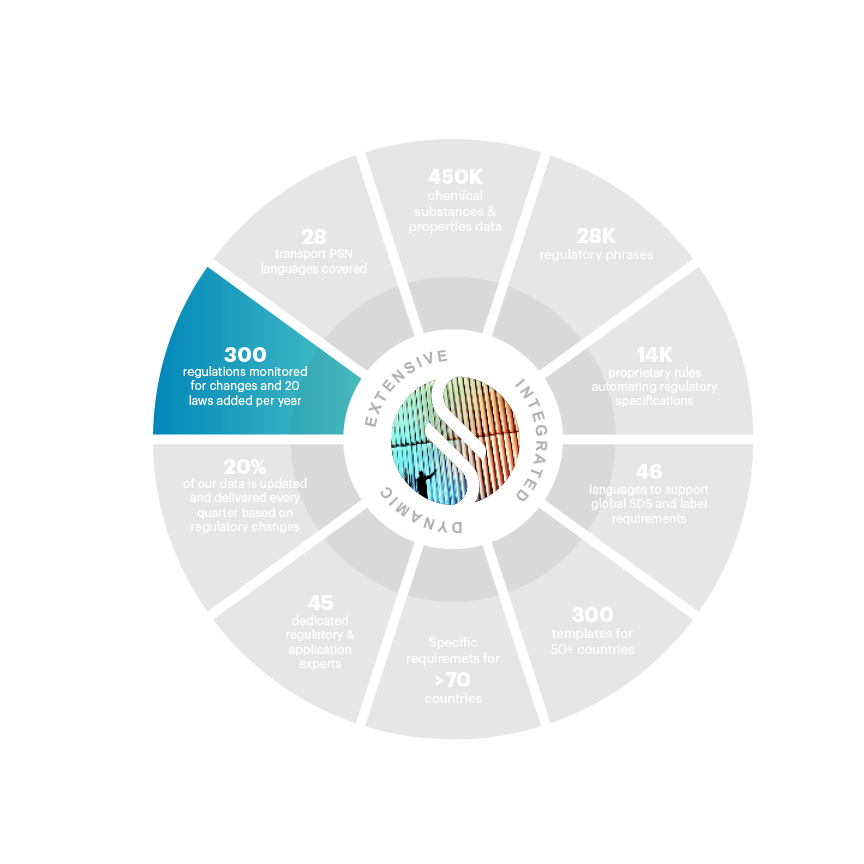
300 regulations monitored
Hazard communication
Industry specific
Chemical control
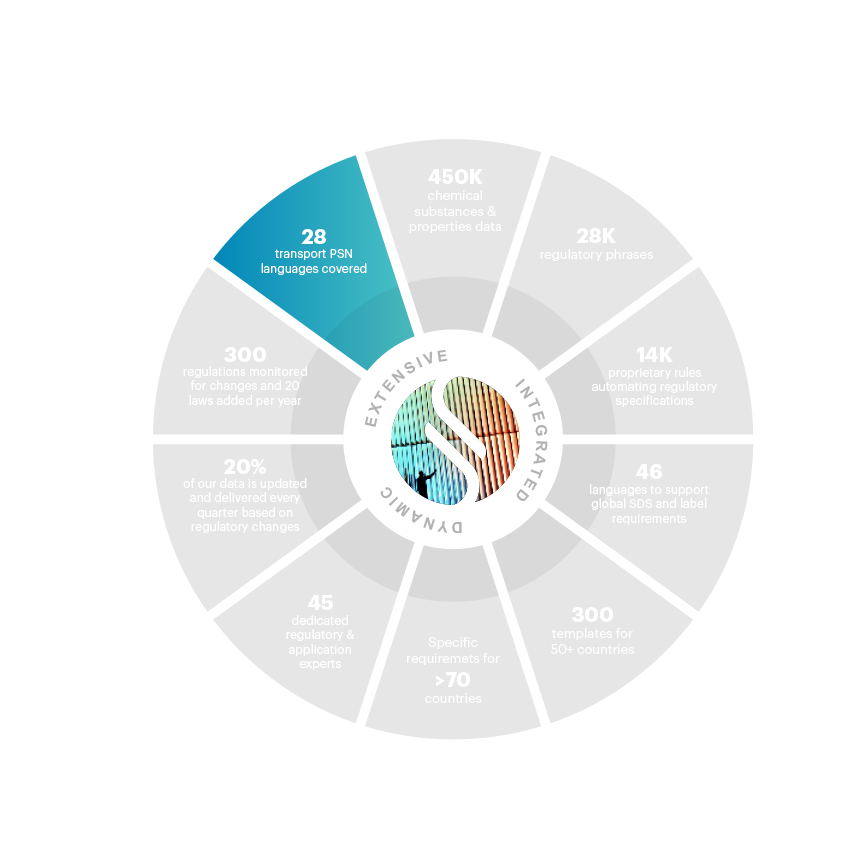
28 PSN languages covered
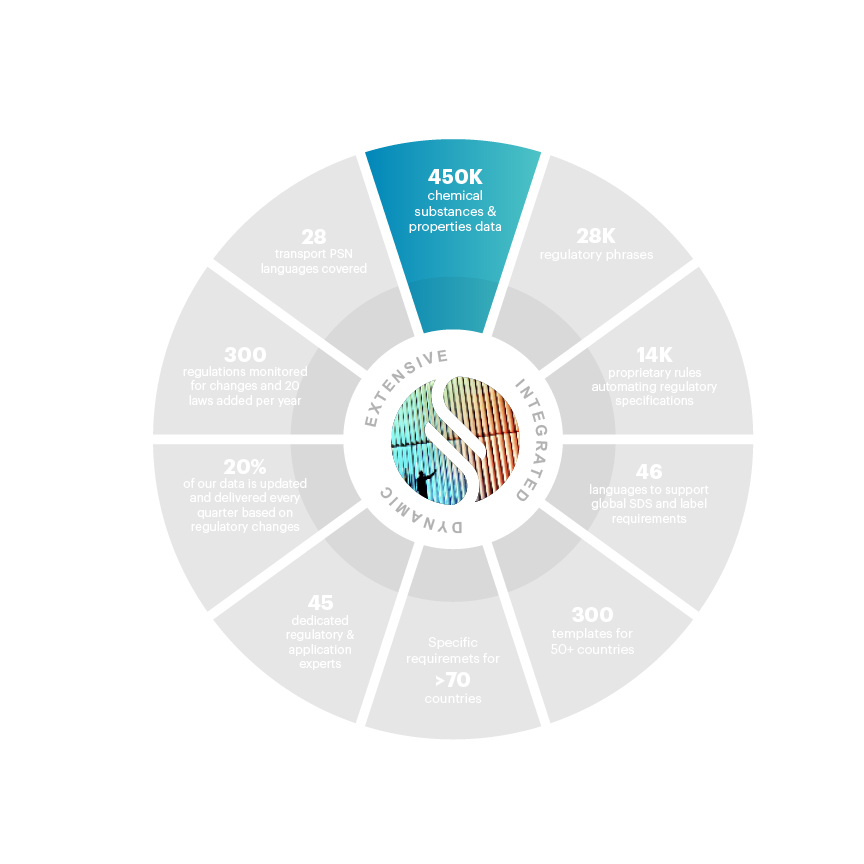
450,000+ chemical substances & properties data
Fill out the form and we will get back to you to discuss your regulatory content needs.


